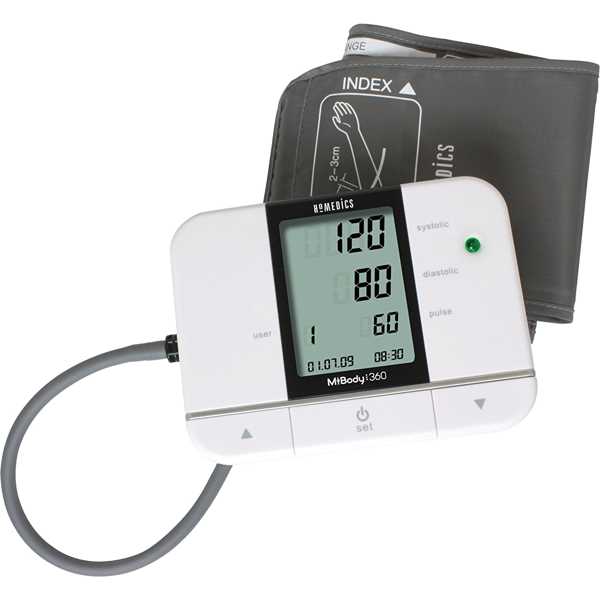
HoMedics Mibody 360EU

Device Name:
Mibody
Device Model:
360EU
Manufacturer:
OBL: HoMedics Inc., 3000 Pontiac Trail, Commerce Township, MI 48390, UNITED STATES.
OEM: Oregon Scientific Inc., 10778 SW Manhasset Drive, Tualatin, OR 97062, UNITED STATES.
Measuring functions:
Blood Pressure and PR
Primary Client Use:
Intended for self-measurement and home use
Measurement Site:
Upper Arm
Measurement Occurrence:
Single measurements only
Availability:
Discontinued but still available
Device Manual:
Description:
The HoMedics Mibody 360EU is an automatic blood pressure monitor. The accuracy of its blood pressure measurement technology has yet to be proven to MDR requirements. Blood pressure measurements are taken from the upper arm. It is intended for self-measurement and home use. This device has been discontinued by the manufacturer but may be available through certain outlets.
Assessment:
While the technology used in the HoMedics Mibody 360EU, to measure blood pressure, has been declared as being equivalent to that used in a clinically validated device, no evidence has been published to show that the devices have been compared according to a protocol compliant with (EU) 2017/745 and MEDDEV 2.7/1 rev 4.
Recommendations:
| Accuracy Assessment | Recommendation | Basis | |
| BP | Medaval | None | Although equivalence to another device is claimed, this has not been tested to MDR requirements. |
| BP | BIHS (UK and IRL) | Self-measurement | Manufacturer declaration of equivalence to the Oregon BPU 330 |
| BP | ESH (Europe) | Self-measurement | The BIHS recommendation, despite the absence, at the time, of the scientific evidence required by the same authors' criteria. |
Device Family:
HoMedics Mibody 360EUB, Oregon BPU 330B, Oregon Ssmart Talking (BPU321OS)B, Oregon Tunstall BPU-329B
Legend: B BIHS Derivative
Legend: B BIHS Derivative
Validation Publications for Equivalent Devices:
Note: This is a provisional list, as equivalence according to EU Regulation 2017/745 (e.g. MEDDEV 2.7/1 rev 4) is not proven. Accordingly, these publications are not used in the assessment of star-ratings.
Oregon BPU 330
Li L, Zhang X, Yan C, Liang Q. Validation of the Oregon Scientific BPU 330 for self-monitoring of blood pressure according to the International Protocol. Vasc Health Risk Manag. 2008;4(5):1121-5. PMID: 19183762. Available from: PMC2605325.
ESH-IP:2002 - Pass General population
