
OBL: B. Braun Melsungen AG, Carl-Braun-Straße, 134212 Melsungen, GERMANY.
OEM: ShenZhen Kingyield Technology Co. Ltd., Building C, Fuhai Industrial Zone, Fuhai Road, FuYong Town, BaoAn, Shenzhen, GuangDong 518103, CHINA.
| Accuracy Assessment | Recommendation | Basis | |
| BP | Medaval | None | Although equivalence to another device is claimed, this has not been tested to MDR requirements. |
Legend: D DET Equivalence, F FDA 510(k)
Note: This is a provisional list, as equivalence according to EU Regulation 2017/745 (e.g. MEDDEV 2.7/1 rev 4) is not proven. Accordingly, these publications are not used in the assessment of star-ratings.
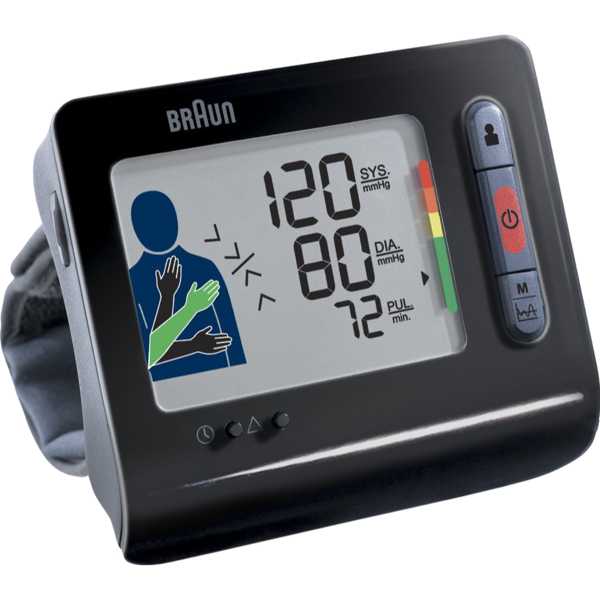
Kingyield BP-210
Zeng WF, Huang QF, Sheng CS, Li Y, Wang JG. Validation of the Kingyield BP210 wrist blood pressure monitor for home blood pressure monitoring according to the European Society of Hypertension-International Protocol. Blood Press Monit. 2012 Feb;17(1):42-4. doi: 10.1097/MBP.0b013e32834f8301. PMID: 22185959.
ESH-IP:2002 - Pass General population