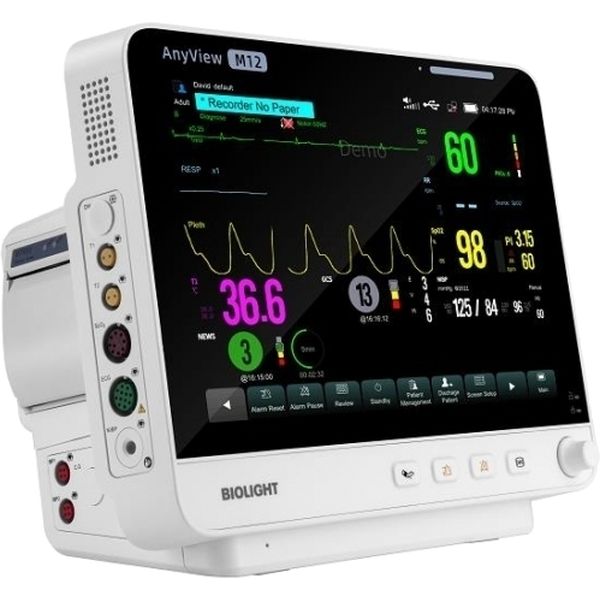
Biolight AnyView M12

Device Name:
AnyView
Device Model:
M12
Manufacturer:
Guangdong Biolight Meditech Co. Ltd., 2 Innovation First Road, Technical Innovation Coast, Hi-tech Zone, Zhuhai, CHINA.
Measuring functions:
Patient monitor
Primary Client Use:
Intended for patient monitoring
Measurement Site:
BP: Upper Arm; SpO2: Finger
Measurement Occurrence:
BP: Single and intermittent measurements; SpO2: Single, intermittent and continuous measurements
Availability:
Available Currently
Description:
The Biolight AnyView M12 is a patient monitor. Medaval has not found evidence proving the accuracy of its blood pressure measurement technology and information regarding the accuracy of its oxygen saturation measurement technology does not appear to be available. Blood pressure measurements are taken from the upper arm and oxygen saturation measurements are taken from the finger. It is intended for bedside patient monitoring.
Assessment:
There appears to be no peer-reviewed clinical validation information available on the technology used in the Biolight AnyView M12 either to measure blood pressure or to measure peripheral oxygen saturation.
Recommendations:
| Accuracy Assessment | Recommendation | Basis | |
| BP | Medaval | Not recommended | This device has not been clinically validated. |