| Accuracy Assessment | Recommendation | Basis | |
| BP | Medaval | None | Although equivalence to another device is claimed, this has not been tested to MDR requirements. |
| BP | BIHS (UK and IRL) | Self-measurement | Manufacturer declaration of equivalence to the A&D UA-651 |
| Consumer Body | Year(s) | Level | Comment | |
| BP | Choice (Australia) | Latest: 2019-2022 | A |
Legend: B BIHS Derivative, J JHS Implied Equivalence, P Stride Equivalence, Z Almost Certain
Note: This is a provisional list, as equivalence according to EU Regulation 2017/745 (e.g. MEDDEV 2.7/1 rev 4) is not proven. Accordingly, these publications are not used in the assessment of star-ratings.
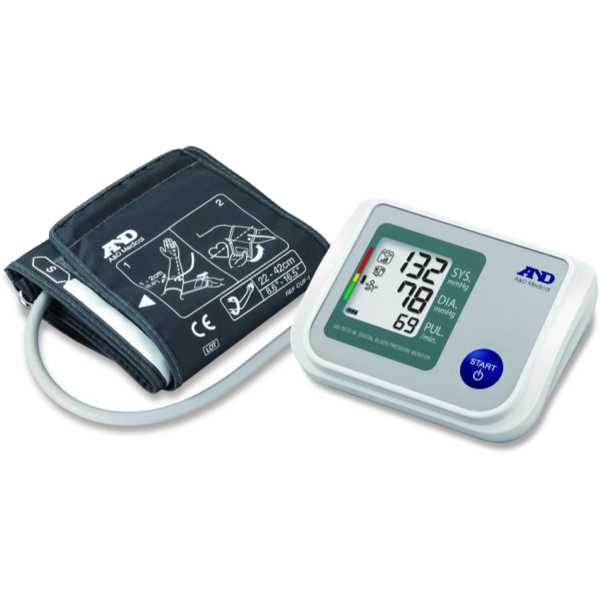
A&D UA-651
Benetti E, Fania C, Palatini P. Validation of the A&D BP UA-651 device for home blood pressure measurement according to the European Society of Hypertension International Protocol revision 2010. Blood Press Monit. 2014 Feb;19(1):50-3. doi: 10.1097/MBP.0000000000000020. PMID: 24346435.
ESH-IP:2010 - Pass General population
Benetti E, Fania C, Palatini P. Validation of the A&D BP UA-651 device with a wide-range cuff for home blood pressure measurement according to the European Society of Hypertension International Protocol revision 2010. Blood Press Monit. 2015 Jun;20(3):164-7. Epub: 2014 Dec 22. doi: 10.1097/MBP.0000000000000100. PMID: 25536400.
ESH-IP:2010 - Pass General population